| Crystal Structure Prediction and its Application in Earth and Materials Sciences |
 )
) at ambient condition
at ambient condition To confirm the validity of our methods, we first explore the energy lanscape of Mg(BH )
) at ambient condition. The crystal structure of Mg(BH
at ambient condition. The crystal structure of Mg(BH )
) at ambient condition has been extensively studied as a template for developing novel hydrogen-storage solutions. Based on the experimental data, the ground state
at ambient condition has been extensively studied as a template for developing novel hydrogen-storage solutions. Based on the experimental data, the ground state  and
and 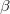 phases, have been assigned space groups
phases, have been assigned space groups 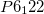 (330 atoms per unit cell) and
(330 atoms per unit cell) and 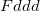 (704 atoms/cell), which have unexpectedly complex crystal structures 128; 129; 130; 131; 132. There had been disputes between experimentalists and theoreticians regarding the nature of the ground-state structure of Mg(BH
(704 atoms/cell), which have unexpectedly complex crystal structures 128; 129; 130; 131; 132. There had been disputes between experimentalists and theoreticians regarding the nature of the ground-state structure of Mg(BH )
) 126; 133; 134; 135. Recent theoretical work then predicted a new body-centered tetragonal phase (with I-4m2 symmetry), which has slightly lower energy than P6
126; 133; 134; 135. Recent theoretical work then predicted a new body-centered tetragonal phase (with I-4m2 symmetry), which has slightly lower energy than P6 phase, by using the prototype electrostatic ground-state approach (PEGS) 133. Later, based on the prototype structure of Zr(BH
phase, by using the prototype electrostatic ground-state approach (PEGS) 133. Later, based on the prototype structure of Zr(BH )
) , another orthorhombic phase with F222 symmetry was found to have even lower energy than all previously proposed structures 135.
, another orthorhombic phase with F222 symmetry was found to have even lower energy than all previously proposed structures 135.
![\includegraphics[scale=0.4]{chapter4/pdf/Fig-MgBH.png}](images/img-0227.png)
 )
) polymorphs at ambient conditions found by USPEX. a) F222 phase; b) I4
polymorphs at ambient conditions found by USPEX. a) F222 phase; b) I4 22 phase.
22 phase. In general, the previous theoretical discoveries of novel Mg(BH )
) phases were conducted either by ad hoc extensive searching or by chemical intuition. However, our evolutionary algorithm does not rely on any prior knowledge except chemical composition, and could be particularly useful for predicting stable crystal structures for these complex metal hydride systems. If we consider the BH
phases were conducted either by ad hoc extensive searching or by chemical intuition. However, our evolutionary algorithm does not rely on any prior knowledge except chemical composition, and could be particularly useful for predicting stable crystal structures for these complex metal hydride systems. If we consider the BH
 ion as a molecular group, the search space would be dramatically reduced. Within 10 generations
ion as a molecular group, the search space would be dramatically reduced. Within 10 generations  400 structure relaxations, USPEX found the F222 phase (Fig. 4.1a) as the most stable structure at ambient pressure. Meanwhile, I-4m2 (Fig. 4.1b) was also found by USPEX in the same calculation, with enthalpy less than 1.2 meV/atom above that of F222 phase. Compared to the previous work, our method is clearly more universal and robust, enables efficient structure prediction for complex molecular systems, both organic and inorganic.
400 structure relaxations, USPEX found the F222 phase (Fig. 4.1a) as the most stable structure at ambient pressure. Meanwhile, I-4m2 (Fig. 4.1b) was also found by USPEX in the same calculation, with enthalpy less than 1.2 meV/atom above that of F222 phase. Compared to the previous work, our method is clearly more universal and robust, enables efficient structure prediction for complex molecular systems, both organic and inorganic.
![\includegraphics[scale=1.0]{chapter4/pdf/Fig1.png}](images/img-0228.png)
 )
) polymorphs at high pressures found by USPEX. (a), (c) Projections of the
polymorphs at high pressures found by USPEX. (a), (c) Projections of the  structure along [001] and [010] directions, (b), (d) projections of the
structure along [001] and [010] directions, (b), (d) projections of the  structure along [001] and [010] directions. Blue, red, and green spheres represent Mg, B, and H atoms, respectively.
structure along [001] and [010] directions. Blue, red, and green spheres represent Mg, B, and H atoms, respectively.