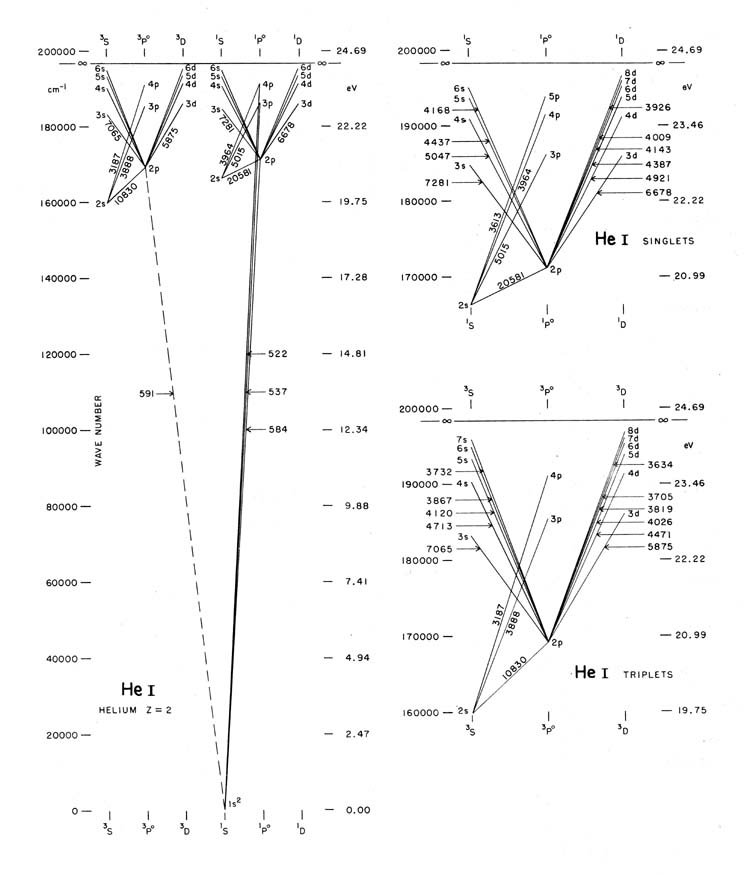
Caption: Grotrian diagram of He I (i.e., neutral helium).
Features:
- The labeled
atomic line
wavelengths are in
angstroms (Å).
- Helium is a
noble gas, and so has tightly bound
electrons.
Thus, exciting helium to even the lowest-energy excited states (i.e., energy levels of energy above the ground state) takes considerable energy supplied by either or both of heat energy and photon energy.
- The diagram shows that even the 1st
excited-state
energy level is
∼ 19.75 electron-volts (eV) above
the ground state.
Note that 19.75 eV means that high occupation of this energy level in thermodynamic equilibrium conditions requires a temperature of >∼ 2*10**5 K.
The argument is as follows. The fractional occupation of an excited state is of order the Boltzmann factor exp[-E/(kT)], where E is the energy above the ground state, T is temperature, and Boltzmann contant k = 8.617 3303(50)*10**(-5) eV/K ≅ 10**(-4) eV/K (see NIST: CODATA: Constants: Complete Listing). So for the Boltzmann factor to be >∼ exp(-1) one must have E/(kT) <∼ 1, and so T >∼ 2*10**5 K.
- Most atoms have some much
lower energy
excited states
than He I.
Those excited states are, in fact, more abundantly occupied in stars with relatively low (stellar atmosphere) temperatures than the excited states of He I.
- Typically,
He I
absorption lines
are strong
for
B stars
with temperatures
∼ 11000--25000 K
(see UCL: The Classification of Stellar Spectra) where
the relative occupation of the
excited states is still
<< exp(-1), but evidentlly
still big enough for relatively strong
absorption lines.
Very weak He I absorption lines can be found in lower temperature stars (e.g., the Sun) if you look in detail (see Wikipedia; Fraunhofer lines: Naming).
- The most prominent
He I
absorption line
in the visible band
in stellar spectra
is usually the
He I 5876 Å line
(which is labeled by 5875 in the figure).
- The ultraviolet
absorption lines
arising from the ground state
are unobservable by
ground-based astronomy
because of the strong
opacity
of the Earth's atmosphere
in the ultraviolet
blueward of
∼ 3000 Å
(see Wikipedia:
Earth's atmosphere: Absorption).
-
As you go to hotter stars than
B2 stars,
He I
absorption lines
get weaker.
This is because as temperature increases,
the helium becomes
increasingly ionized
i.e., becomes increasingly singly-ionized
He II which exhibits
He II
spectral lines.
- For O stars,
He II
absorption lines
and/or emission lines
are the dominant
helium
spectral lines
(see UCL: The Classification of Stellar Spectra).
- Actually,
absorption lines
of any kind of
helium
are only ever strong in
stars
because helium
is so abundant---about 25 % by
mass fraction
of baryonic matter in the
observable universe.
- Note that non-thermal processes can lead to strong
helium
spectral lines
(absorption lines
or emission lines)
when low temperature would otherwise
preclude them.
Local file: local link: grotrian_02_00_He_I.html.
File: Grotrian diagram file: grotrian_02_00_He_I.html.